
By substituting deuterium atoms for hydrogen atoms, scattering noise is reduced. Furthermore, conventional hydrogen has a high incoherent neutron cross section, which is nil for D, which is a nuisance problem. The availability of deuterated samples benefits neutron scattering methods in particular: The H and D cross sections are extremely distinct and have opposite signs, allowing for contrast change in these studies. In grams, it is the mass of 1 mole of the material, or $ 6.022\times H $ ) (deuterium has a mass of 2.014102 u, compared to the mean hydrogen atomic weight of 1.007947 u and protium's mass of 1.007825 u) confers non-negligible chemical differences with protium-containing compounds, whereas isotope weight ratios within other chemical elements are largely insignificant in this regard). It also describes how these concepts apply to the work that the Department of Energy’s Office of Science conducts as it helps the United States excel in research across the scientific spectrum.Hint: The mass of a sample of a chemical compound divided by the amount of material in that sample, measured in moles, is the molar mass of that compound in chemistry. DOE Explains offers straightforward explanations of key words and concepts in fundamental science. The recently launched Facility for Rare Isotope Beams has completed its groundbreaking first experimental results.National Isotope Development Center ( Isotope Basics).DOE Isotope Development & Production for Research and Applications.The Journey of Actinium-225: How Scientists Discovered a New Way to Produce a Rare Medical Radioisotope. 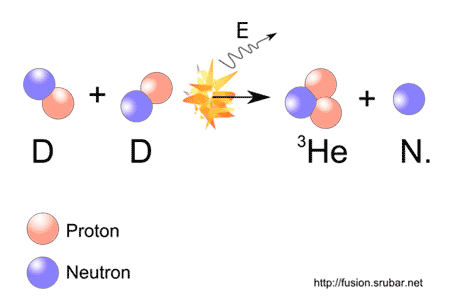
NSAC: Compelling Research Opportunities Using Isotopes.NSAC Report: Meeting Isotope Needs and Capturing Opportunities for the Future.Hydrogen is the only element whose isotopes have unique names: deuterium for hydrogen with one neutron and tritium for hydrogen with two neutrons.Some elements can only exist in an unstable form (for example, uranium).All artificial (lab-made) isotopes are unstable and therefore radioactive scientists call them radioisotopes.Take the molecular weight of water to be 18. Keep in mind the atomic weights of hydrogen and deuterium are approximately 1 and 2, respectively. There are two main types of isotopes: stable and unstable (radioactive). Transcribed image text: Estimate the mass (in kg) of deuterium in an 80,000- L swimming pool, given the deuterium makes up 0.0150 of natural hydrogen atoms.Finally, it conducts research and development on new and improved isotope production and processing techniques. The program also maintains the infrastructure required to produce and supply priority isotope products and related services. The program produces and distributes radioactive and stable isotopes that are in short supply, including byproducts, surplus materials, and related isotope services. The DOE Isotope Program addresses this need. However, isotopes are not always available in sufficient quantities or at reasonable prices. Isotopes are needed for research, commerce, medical diagnostics and treatment, and national security. They are important in nuclear medicine, oil and gas exploration, basic research, and national security. Isotopes have unique properties, and these properties make them useful in diagnostics and treatment applications. Generally the value of a tracer arises from the fact that, although its difference in mass or its radioactivity permits its detection, it is essentially active in the same way that the ordinary atoms of the element are. This decay means the amount of carbon-14 in an object serves as a clock, showing the object’s age in a process called “carbon dating.” Both deuterium and tritium are useful as isotopic tracers for the investigation of chemical structures and of reaction mechanisms. Carbon-14 is unstable and undergoes radioactive decay with a half-life of about 5,730 years (meaning that half of the material will be gone after 5,730 years). Carbon-12 is stable, meaning it never undergoes radioactive decay. The addition of even one neutron can dramatically change an isotope’s properties. Every element has its own number of isotopes. Carbon occurs naturally in three isotopes: carbon 12, which has 6 neutrons (plus 6 protons equals 12), carbon 13, which has 7 neutrons, and carbon 14, which has 8 neutrons. For example, carbon has six protons and is atomic number 6. The number of protons in a nucleus determines the element’s atomic number on the Periodic Table. Isotopes are members of a family of an element that all have the same number of protons but different numbers of neutrons. Elements have families as well, known as isotopes. A family of people often consists of related but not identical individuals. The biennial review of atomic-weight determinations and other cognate data has resulted in changes for the standard atomic weights of 19 elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
